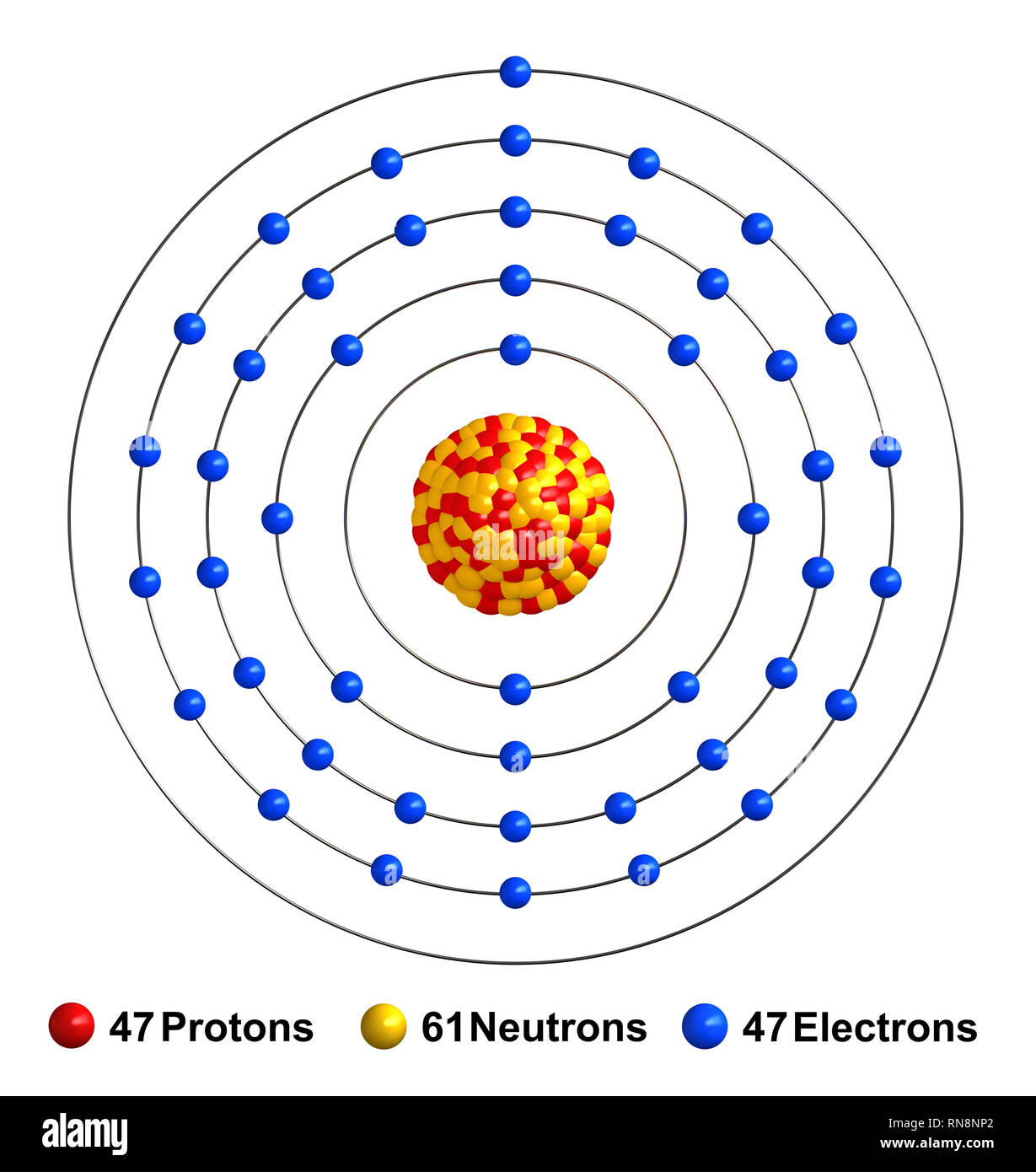 
We will need 2 aluminum atoms to get a +6 charge and 3 oxygen atoms to get a -6 charge. Oxygen #s^2p^4# has 6 valence electrons and an oxidation state of -2 or #O^(−2)# This makes the formula for calcium chloride, #CaCl_2#.Īluminum #s^2p^1# has 3 valence electrons and an oxidation state of +3 or #Al^(+3)# This means that two #Cl^(−1)# anions will balance with one #Ca^(+2)# cation. 
Ionic bonds form when the charges between the metal cation and non-metal anion are equal and opposite. It needs one electron to make it stable at 8 electrons in its valence shells. This makes calcium a Ca+2 cation.Ĭhlorine is a Halogen in the 17th column or #s^2p^5# group.Ĭhlorine has 7 valence electrons. This means that calcium #s^2# has 2 valence electrons it readily gives away in order to seek the stability of the octet. Lets take the ionic formula for Calcium Chloride is #CaCl_2#Ĭalcium is an Alkaline Earth Metal in the second column of the periodic table. Phosphorus has 5 valence electrons 2 from the 3s and 3 from the 3p Sodium has 1 valence electron from the 3s orbital These electrons are found in the s and p orbitals of the highest energy level for the element. Because its 2n shell is filled, it is energetically stable as a single atom and will rarely form chemical bonds with other atoms.The valence electrons are the electrons that determine the most typical bonding patterns for an element. For instance, lithium ( Li \text Ne start text, N, e, end text ), on the other hand, has a total of ten electrons: two are in its innermost 1 s 1s 1 s 1, s orbital and eight fill the second shell-two each in the 2 s 2s 2 s 2, s and three p p p p orbitals, 1 s 2 1s^ 2 1 s 2 1, s, squared 2 s 2 2s^ 2 2 s 2 2, s, squared 2 p 6 2p^6 2 p 6 2, p, start superscript, 6, end superscript. Elements in the second row of the periodic table place their electrons in the 2n shell as well as the 1n shell. After the 1 s 1s 1 s 1, s orbital is filled, the second electron shell begins to fill, with electrons going first into the 2 s 2s 2 s 2, s orbital and then into the three p p p p orbitals. The second electron shell, 2n, contains another spherical s s s s orbital plus three dumbbell-shaped p p p p orbitals, each of which can hold two electrons. Hydrogen and helium are the only two elements that have electrons exclusively in the 1 s 1s 1 s 1, s orbital in their neutral, non-charged, state. On the periodic table, hydrogen and helium are the only two elements in the first row, or period, which reflects that they only have electrons in their first shell. This is written out as 1 s 2 1s^ 2 1 s 2 1, s, squared, referring to the two electrons of helium in the 1 s 1s 1 s 1, s orbital. 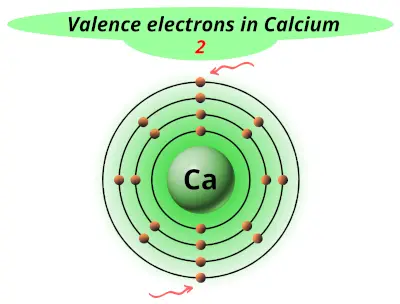
Helium has two electrons, so it can completely fill the 1 s 1s 1 s 1, s orbital with its two electrons. This can be written out in a shorthand form called an electron configuration as 1 s 1 1s^ 1 1 s 1 1, s, start superscript, 1, end superscript, where the superscripted 1 refers to the one electron in the 1 s 1s 1 s 1, s orbital. Hydrogen has just one electron, so it has a single spot in the 1 s 1s 1 s 1, s orbital occupied. The 1 s 1s 1 s 1, s orbital is the closest orbital to the nucleus, and it fills with electrons first, before any other orbital. The first electron shell, 1n, corresponds to a single 1 s 1s 1 s 1, s orbital.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
